38% of Indian adults have fatty liver disease. This is not a pre-disease or borderline condition. It is clinically confirmed Metabolically Dysregulated Steatotic Liver Disease (MASLD).
MASLD builds up silently in the liver. It drives insulin resistance and increases cardiovascular risk. Over time, it can progress to fibrosis, cirrhosis, and even hepatocellular carcinoma. However, most patients do not feel any symptoms.
The most alarming issue is not just its high prevalence. Instead, it is how we assess it. Standard tools like liver ultrasound, ALT, and BMI often miss the metabolic drivers behind the disease.
India’s MASLD burden is especially severe. The “thin-fat” phenotype is common in Indian adults. This means people may have high visceral fat and low skeletal muscle, even at a normal BMI.
As a result, MASLD develops at lower thresholds in Indian patients. It also progresses faster, especially with diabetes and insulin resistance. At the same time, BMI-based screening fails to detect it early.
Body composition technology is changing this reality. In particular, tools like InBody BIA provide deeper metabolic insights. In this article, we explore seven proven ways InBody BIA is transforming MASLD detection, staging, and management in India.
| 38% MASLD prevalence in Indian adults estimated by INASL (Indian National Association for Study of the Liver) | 19.28% MASLD prevalence in normal-weight (BMI below 25) individuals lean MASLD invisible to BMI screening (PMC 2025) | 55%+ Projected global adult MASLD prevalence by 2040, India bears the highest disease burden among GBD 2021 nations | 5 min Time for InBody to deliver visceral fat, muscle mass, ECW/TBW, and phase angle the complete MASLD metabolic picture |
Why Standard Assessment Is Failing India’s MASLD Patients
Liver ultrasound detects hepatic steatosis but only when fat accumulation exceeds approximately 20–30% of hepatocytes. It provides no information about visceral fat distribution, skeletal muscle mass, or the metabolic drivers of the disease. ALT is unreliable; it can be normal in up to 79% of patients with biopsy-proven NASH. BMI in Indian patients is a fundamentally flawed screening tool: the India-Specific Consensus on MASLD in Type 2 Diabetes (Diabetes Care, October 2025) explicitly recognises elevated visceral fat burden at lower BMI thresholds in Asian Indians, meaning that standard BMI cutoffs systematically underestimate MASLD risk in this population.
The 2025 PMC study on lean MASLD confirmed this failure in quantitative terms: MASLD was present in 19.28% of normal-weight individuals despite a normal BMI. These patients presented with visceral adiposity as a significant independent predictor of hepatic steatosis (p < 0.001) yet their BMI provided no clinical signal whatsoever. In Indian clinical practice, these patients are not being screened for MASLD. They are being reassured that their weight is healthy.
THE LEAN MASLD PROBLEM IN INDIA
Research consistently shows that MASLD is increasing in India, even among individuals with normal BMI, due to central obesity and genetic factors. Visceral fat, not total body weight, is the primary driver of hepatic fat accumulation in Indian patients. A patient with a BMI of 22.5 and a Visceral Fat Level of 11 measured by InBody has a clinically significant MASLD risk that their BMI completely conceals. This is the metabolic reality that standard assessment tools are missing in millions of Indian patients every year. Learn more about why BMI misses what body composition reveals.
Way 1: Measuring Visceral Fat: The Primary Driver of Hepatic Steatosis
Visceral fat is the primary metabolic driver of MASLD. Visceral adipocytes release free fatty acids into the portal circulation, flooding the liver with lipotoxic substrate. They secrete IL-6, TNF-α, and resisting, inflammatory cytokines that drive hepatic insulin resistance, de novo lipogenesis, and progression from simple steatosis to metabolic dysfunction-associated steatohepatitis (MASH). The Biores Scientia 2025 review of India’s MASLD burden confirmed that visceral fat is a better indicator of liver fat accumulation than overall body weight, making it the single most important modifiable MASLD risk factor.
Waist circumference is an imprecise proxy for visceral fat. Abdominal ultrasound measures hepatic fat, not the visceral fat producing it. BMI measures neither. InBody directly measures Visceral Fat Level (VFL) on a 1–20 scale and Visceral Fat Area (VFA) in cm², enabling hepatologists to quantify the primary driver of their patient’s MASLD, set a specific reduction target, and track its serial response to lifestyle and pharmacological intervention.
CLINICAL IMPLICATION
The India-Specific Consensus on MASLD (Diabetes Care, 2025) mandates routine MASLD screening in all Indian individuals with T2DM using non-invasive tools, and explicitly defines India-specific cutoffs that recognise elevated visceral fat burden at lower BMI. InBody’s direct VFL measurement provides the visceral fat quantification that waist circumference cannot, and enables targeted treatment decisions. SGLT-2 inhibitors, GLP-1 agonists, and resistance training all reduce visceral fat measurably, but only InBody confirms this is occurring in your individual patient.
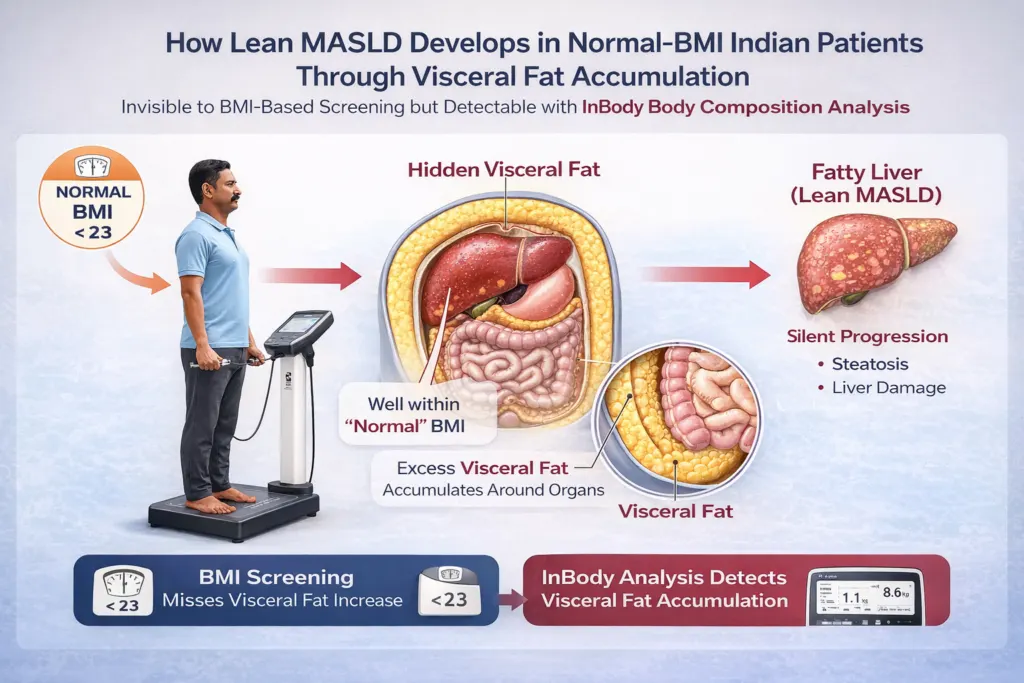
Way 2: Detecting Lean MASLD Before It Progresses to Fibrosis
Lean MASLD fatty liver disease in patients with normal BMI is a uniquely dangerous clinical phenotype because it is systematically missed by BMI-based screening. The 2025 PMC lean MASLD study confirmed that despite normal BMI, this cohort had considerable metabolic dysfunction: hypertension in 26.47%, hypercholesterolaemia in 46.54%, and T2DM in 18.38%. Visceral adiposity captured not by BMI but by body composition analysis was the significant independent predictor of hepatic steatosis (p < 0.001).
For Indian patients, the lean MASLD phenotype is especially prevalent. The 2025 India-Specific MASLD Consensus acknowledges that Indian individuals develop MASLD at BMI levels below 25 kg/m² because of the disproportionately high visceral fat accumulation of the thin-fat phenotype. InBody’s direct visceral fat measurement, combined with body fat percentage both entirely independent of BMI and identifies this high-risk group before hepatic steatosis has progressed to the fibrosis stages that dramatically worsen prognosis.
KEY INSIGHT
The INASL sets the FIB-4 threshold for high-risk MASLD lower in India (score of 1.0 instead of the international 1.3), confirming that Indian patients progress to advanced fibrosis at metabolic risk levels that international guidelines classify as lower risk. Body composition assessment that detects elevated visceral fat and low skeletal muscle mass in apparently healthy-weight Indian patients is the earliest available clinical signal of this accelerated progression trajectory.
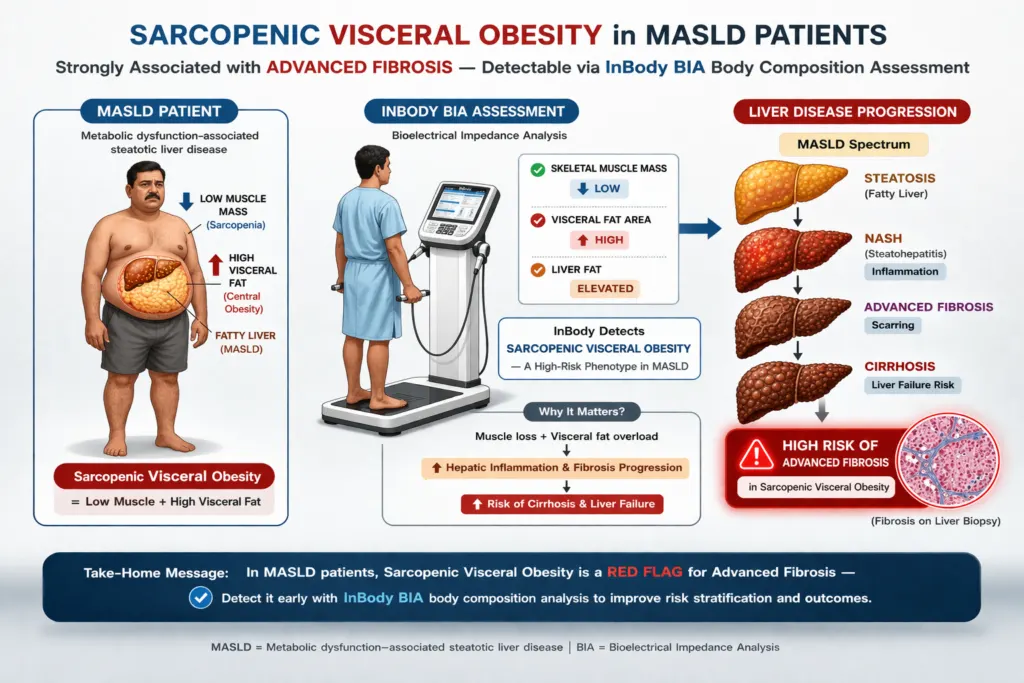
Way 3: Identifying Sarcopenic Visceral Obesity: The Highest-Risk MASLD Phenotype
Sarcopenic visceral obesity (SVO), the concurrent presence of low skeletal muscle mass and excess visceral fat, has emerged as the highest-risk body composition phenotype in MASLD. A landmark 2025 PMC cross-sectional study of 334 adults with MASLD confirmed that SVO was independently associated with advanced liver fibrosis, elevated cardiovascular risk, and significantly worse metabolic markers than either sarcopenia or visceral obesity alone. Sarcopenia was assessed in this study by bioelectrical impedance analysis exactly the technology InBody provides.
The mechanism is dual: visceral fat drives hepatic lipotoxicity and insulin resistance through portal free fatty acid delivery. Skeletal muscle loss simultaneously reduces glucose disposal, worsening systemic insulin resistance and accelerating hepatic de novo lipogenesis. The two processes compound each other. Together they produce a metabolic environment in which MASLD progresses to MASH and fibrosis significantly faster than in patients with either condition alone.
THE SVO CLINICAL IMPLICATION
A MASLD patient who loses weight through caloric restriction without resistance training may reduce total body weight, but lose muscle primarily, leaving visceral fat relatively preserved. Their SVO worsens even as their weight falls. InBody’s simultaneous measurement of Visceral Fat Level and Skeletal Muscle Mass at every visit detects this dangerous phenotypic shift, enabling clinicians to intervene with resistance training and protein supplementation before fibrosis accelerates.
Way 4: Using Phase Angle to Track Fibrosis Progression Non-Invasively
Phase angle, a BIA-derived marker of cellular membrane integrity and overall cell health, declines as liver disease progresses. In MASLD patients, advancing fibrosis disrupts hepatocyte membrane integrity, reduces cellular water retention capacity, and produces progressive phase angle deterioration. This makes phase angle a non-invasive, real-time marker of hepatic disease severity measurable at every InBody scan without liver biopsy, elastography, or specialist referral.
Multiple studies in chronic liver disease confirm that phase angle is negatively correlated with fibrosis stage and positively correlated with liver function and nutritional status. In MASLD patients undergoing treatment with lifestyle intervention, SGLT-2 inhibitors, or GLP-1 agonists, a rising phase angle between serial InBody scans provides an early signal of hepatic improvement months before repeat elastography or biopsy can confirm it. A falling phase angle despite intervention provides an early warning of treatment failure or disease progression.
CLINICAL IMPLICATION
Phase angle closes the monitoring gap in MASLD management. Liver biopsy, the gold standard for fibrosis staging, cannot be repeated frequently. Elastography requires specialist equipment. InBody’s phase angle is available at every scan, at every clinic visit, in under 5 minutes, providing a monthly or bimonthly signal of whether MASLD is responding to treatment at the cellular level, between the infrequent gold-standard assessments.
Way 5: Monitoring the ECW/TBW Ratio as a MASLD Inflammation Marker
MASLD is a profoundly inflammatory condition. Visceral fat releases IL-6 and TNF-α. Portal lipotoxicity drives hepatocyte ER stress and NF-κB activation. Systemic chronic low-grade inflammation promotes fluid retention and extracellular water expansion, raising the ECW/TBW ratio as inflammatory oedema accumulates in tissues. In MASLD patients with concurrent metabolic syndrome, developing nephropathy, or cardiac involvement, this ECW/TBW elevation provides an early, measurable signal of systemic inflammatory burden.
InBody measures the ECW/TBW ratio at every scan, providing a non-invasive, quantitative marker of inflammatory fluid redistribution. A normal ECW/TBW ratio is below 0.380. In MASLD patients with active systemic inflammation or progressing toward MASH, this ratio rises before clinical signs of fluid overload appear. Serial monitoring of ECW/TBW in MASLD patients alongside visceral fat level and skeletal muscle mass creates a comprehensive inflammatory trajectory that standard liver function tests and ultrasound cannot provide.
Way 6: Confirming Whether MASLD Treatment Is Producing the Right Body Composition Changes
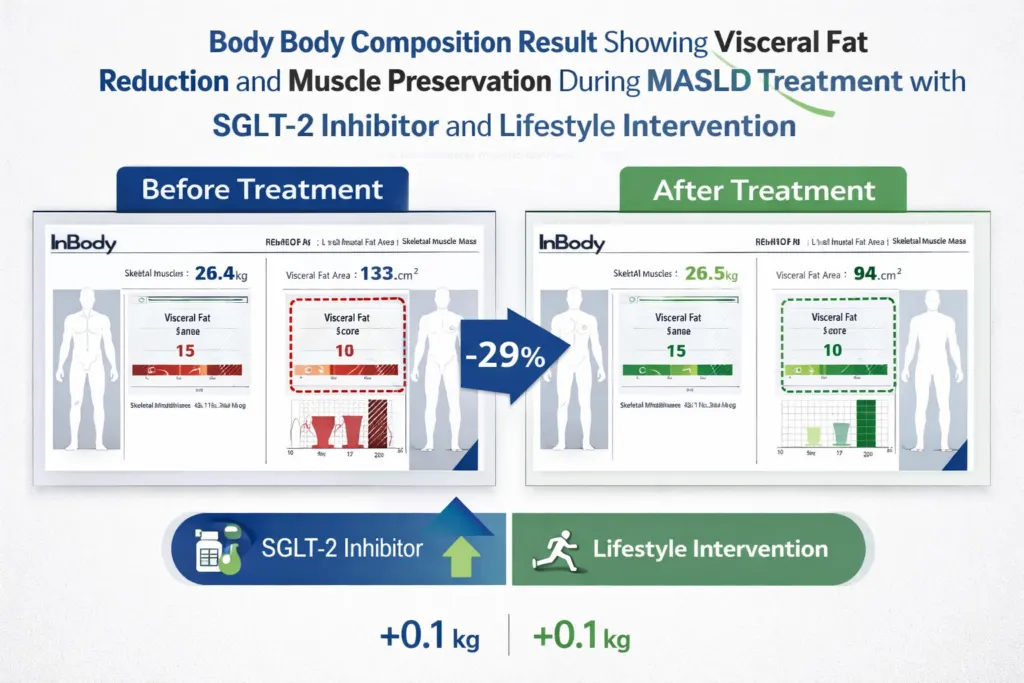
MASLD management in 2026 has multiple evidence-based pharmacological options alongside lifestyle intervention: SGLT-2 inhibitors, GLP-1 receptor agonists, pioglitazone, and for non-cirrhotic MASH with F2–F3 fibrosis resmetirom (the first FDA-approved MASH-targeted medication). The India-Specific MASLD Consensus (Diabetes Care, 2025) prioritises SGLT-2 inhibitors and GLP-1 receptor agonists for Indian patients based on availability, affordability, and dual MASLD and glycaemic benefits.
Every one of these interventions produces body composition changes that can be tracked with InBody, and these body composition changes are detectable at 12 weeks, long before repeat elastography or biopsy can confirm histological response. When a patient on semaglutide loses 8 kg over 16 weeks, InBody reveals whether that weight loss is predominantly visceral fat reduction (a true metabolic win) or includes significant skeletal muscle loss (a concerning signal that requires protein intake adjustment and resistance training). Liver ultrasound alone cannot tell you either.
EVIDENCE
The India-Specific MASLD Consensus mandates routine MASLD screening in all Indian individuals with T2DM using low-cost, non-invasive tools and recommends early use of SGLT2 inhibitors or GLP-1 agonists to prevent fibrosis progression. InBody body composition assessment is a non-invasive tool that confirms whether these interventions are achieving their primary body composition objective: visceral fat reduction alongside muscle preservation. It delivers this confirmation in under 5 minutes at each clinic visit.
Way 7: Motivating Sustained Lifestyle Change: The Most Underestimated MASLD Intervention
Lifestyle intervention, weight loss of 7–10% of body weight through dietary modification and exercise, remains the most effective intervention for reversing hepatic steatosis and preventing fibrosis progression in MASLD. The clinical challenge is not the evidence. It is adherence. Patients on lifestyle modification programmes who cannot see metabolic progress beyond what a scale shows them do not sustain the changes long enough to achieve the 7–10% weight loss needed for histological improvement.
Body composition data closes this adherence gap. When a MASLD patient on a 12-week structured programme sees their InBody result showing Visceral Fat Level reduced from 13 to 10, Skeletal Muscle Mass increased by 1.3 kg, and Body Fat Percentage reduced by 2.8%,, even though their scale weight changed by less than 1.5 kg they have objective, specific, personal evidence that their intervention is working at the metabolic level. That data is more motivating than any coaching, more persuasive than any dietary counselling, and more effective at sustaining adherence than any repeat ultrasound at 6 months.
THE MASLD ADHERENCE PROBLEM AND ITS SOLUTION
Studies consistently confirm that the number-one reason patients with MASLD fail to achieve the 7–10% weight loss needed for histological improvement is loss of motivation within the first 12 weeks. The scale shows modest change. The ultrasound is 6 months away. The ALT barely moved. InBody gives the patient objective proof of visceral fat reduction, muscle gain, and metabolic improvement at 8 and 12 weeks, exactly when adherence most commonly breaks down.
What InBody Measures in MASLD Patients: The Complete Clinical Picture
Each InBody scan in a MASLD patient delivers the following body composition metrics, all in under 5 minutes, without radiation, blood draws, or specialist equipment:
| InBody Metric | What It Measures | Why It Matters in MASLD |
|---|---|---|
| Visceral Fat Level (VFL) | Fat surrounding internal organs 1 to 20 scale | Primary driver of hepatic steatosis and portal lipotoxicity. Target VFL below 10. Reduction confirms treatment response months before repeat ultrasound |
| Visceral Fat Area (VFA) | Visceral fat in cm²: precise quantification | Provides absolute quantification for clinical documentation and serial comparison more precise than waist circumference |
| Skeletal Muscle Mass (SMM) | Lean muscle in kg per limb and trunk | Low SMM = high SVO risk = advanced fibrosis risk. SMM preservation during weight loss distinguishes beneficial fat loss from harmful muscle catabolism |
| Body Fat Percentage | Fat mass as proportion of total body weight | Detects lean MASLD fat metabolic risk invisible to BMI. Essential for identifying the 19%+ of normal-weight patients with significant hepatic steatosis |
| ECW/TBW Ratio | Extracellular to total body water ratio | Marker of MASLD-driven systemic inflammation. Rising ECW/TBW precedes clinical signs of inflammatory fluid overload and tracks treatment response |
| Phase Angle | BIA-derived marker of cellular membrane integrity | Declines with fibrosis progression. Rising phase angle during treatment indicates hepatic improvement months before elastography confirms it |
| InBody Score | Composite body composition health index 0–100 | Single-number tracking of overall metabolic health trajectory is useful for patient communication and treatment target-setting |
Real Case: The MASLD Patient Whose Liver Was Failing Behind a Normal BMI
A 44-year-old female schoolteacher in Pune attended a routine health check organised by her school. Her BMI was 23.4, which falls within the healthy range according to national guidelines. However, a liver ultrasound performed 8 months earlier had shown mild Grade 1 fatty change. At that time, her ALT level was 31 U/L, which remained within the normal range. Based on these findings, her physician recommended dietary modification and noted no urgent concern during the last review.
InBody Scan Reveals Underlying Metabolic Concerns
During the recent health check, an InBody scan revealed a metabolic profile that had not been identified in earlier investigations. The results showed a Visceral Fat Level of 12, crossing the high-risk threshold.
In addition, her Body Fat Percentage was 36.1%, placing her in the obese category despite a normal BMI. Her Skeletal Muscle Mass was 2.3 kg below the expected norm for her age and height.
Meanwhile, her Phase Angle measured 4.6°, falling below the level associated with healthy cellular function. The ECW/TBW ratio was 0.383, indicating a borderline elevation. Furthermore, her Visceral Fat Area was 108 cm², exceeding the 100 cm² threshold linked to higher cardiovascular risk in SVO classification studies.
Intervention Plan and Clinical Approach
Following these findings, her gastroenterologist initiated a structured intervention plan. This included resistance training three times per week and an increase in protein intake to 1.2 g/kg/day. In addition, an SGLT-2 inhibitor was introduced as part of her medical management.
Alongside these measures, she was advised to follow a Mediterranean-style diet with reduced refined carbohydrates.
| ↓3 units Visceral Fat Level (12 → 9 — below high-risk threshold) | +1.4 kg Skeletal Muscle Mass gained | ↑0.3° Phase Angle improvement (4.6° → 4.9°) | ↓2.4% Body Fat Percentage reduced |
Her scale weight changed by 1.2 kg, while her BMI remained at 23.1, still within the “healthy” range. Meanwhile, her ALT dropped to 22 U/L. According to her gastroenterologist, the most clinically significant improvements were the reduction in visceral fat level (VFL) below 10, an increase in phase angle, and gains in muscle mass. These changes were detectable only through an InBody body composition assessment.
Without this data, her Grade 1 fatty liver would likely have been monitored annually using ultrasound alone. As a result, her metabolic progression toward SVO and advanced fibrosis might have continued unnoticed.
Frequently Asked Questions
Q. How common is fatty liver disease in India?
The INASL estimates MASLD prevalence at 38% in Indian adults and 35% in children, one of the highest rates globally. The ICOM-D cohort confirmed a pooled NAFLD/MASLD prevalence of 38.6% in India. Global MASLD prevalence is projected to exceed 55% of adults by 2040, and India bears the highest disease burden among the three nations in the GBD 2021 study. India is home to one of the highest absolute numbers of MASLD patients worldwide, compounded by the lean MASLD phenotype that makes BMI-based screening inadequate.
Q. What body composition metrics matter most in fatty liver disease?
The most clinically relevant InBody metrics in MASLD are: Visceral Fat Level (VFL) primary driver of hepatic steatosis; Skeletal Muscle Mass (SMM), low SMM predicts advanced fibrosis through SVO phenotype; Body Fat Percentage detects lean MASLD invisible to BMI; ECW/TBW ratio, elevated in MASLD due to systemic inflammation; and Phase Angle declines with fibrosis progression, rises with treatment response. These five metrics together provide the complete MASLD metabolic picture that liver ultrasound, ALT, and BMI alone cannot deliver.
Q. How does InBody help monitor fatty liver disease treatment response?
MASLD management lifestyle intervention, SGLT-2 inhibitors, GLP-1 agonists, pioglitazone, and resmetiro produce body composition changes that InBody detects at 12 weeks, long before a repeat liver biopsy or elastography can confirm histological response. InBody serial assessment confirms whether visceral fat level is declining, skeletal muscle is being preserved during weight loss, phase angle is rising (indicating hepatic improvement), and ECW/TBW ratio is normalising. The India-Specific MASLD Consensus recommends low-cost non-invasive tools alongside ultrasound for MASLD monitoring. InBody is the non-invasive tool that delivers these body composition endpoints.
Key Takeaways
- 38% of Indian adults have MASLD, with India bearing the highest disease burden in the GBD 2021 study. Global prevalence is projected to exceed 55% by 2040. BMI-based screening is systematically missing this burden in the Indian population.
- 19.28% of normal-weight individuals have MASLD, lean MASLD driven by visceral fat accumulation, not total body weight. Only InBody’s direct visceral fat measurement and body fat percentage identify this high-risk group before fibrosis develops.
- Sarcopenic visceral obesity (SVO) detected by InBody’s simultaneous VFL and SMM measurement is independently associated with advanced liver fibrosis in MASLD patients. A patient losing weight without resistance training may be worsening their SVO even as their scale weight falls.
- Phase angle declines with fibrosis progression and rises with treatment response, providing a monthly non-invasive signal of hepatic health that bridges the long gaps between elastography and biopsy assessments.
- The India-Specific Consensus on MASLD (Diabetes Care, 2025) prioritises SGLT-2 inhibitors and GLP-1 agonists for Indian patients. InBody is the tool that confirms whether these interventions are achieving their primary objective: visceral fat reduction alongside muscle preservation at 12 weeks, not 12 months.
- InBody delivers all seven MASLD-relevant body composition metrics: VFL, VFA, SMM, body fat %, ECW/TBW ratio, phase angle, and InBody Score in under 5 minutes, at every clinic visit, without radiation, blood draws, or specialist equipment.
Detect the Visceral Fat and Muscle Loss That Liver Ultrasound Cannot See
InBody body composition assessment delivers the visceral fat, muscle mass, phase angle, and inflammation data that complete the clinical picture of MASLD in under 5 minutes, at every hepatology and metabolic clinic visit, in India.

Find an InBody Scanner Near You
References & Further Reading
- GBD 2021 analysis: MASLD burden in India, China, US — India bears highest burden. PMC12482587. 2025.
- ICOM-D cohort: Pooled NAFLD/MASLD prevalence 38.6% in India. Journal of Hepatology. 2024.
- Lean MASLD in normal-weight individuals (BMI <25): 19.28% prevalence, visceral adiposity as predictor. PMC12317428. 2025.
- Sarcopenic visceral obesity in 334 MASLD patients — BIA-assessed sarcopenia, advanced fibrosis risk. PMC12540545. 2025.
- India-Specific Consensus on MASLD in Type 2 Diabetes — SGLT-2, GLP-1, India-specific cutoffs. Diabetes Care. October 2025.
- Chakrabarti SK. India’s Hidden Liver Crisis — visceral fat, lean MASLD, thin-fat phenotype. Biores Scientia. 2025.
- Global Consensus Recommendations for MASLD and MASH — INASL 38% prevalence, FIB-4 India cutoff 1.0. Gastroenterology. 2025.
- Kalra S et al. Indian Consensus on Sarcopenia including MASLD recommendations. Int J Gen Med. 2025;18:1731–1745.




