Health 10 min read
Semaglutide & Muscle Loss: What GLP-1 Drugs Do to Your Body
The Problem: Weight Doesn’t Tell the Whole Story GLP-1 therapy conversations typically focus on blood sugar control and body weight reduction. This simplified framework misses what actually governs…
Reading about body composition? Find an InBody test centre near you →
The Problem: Weight Doesn’t Tell the Whole Story
GLP-1 therapy conversations typically focus on blood sugar control and body weight reduction. This simplified framework misses what actually governs how your body responds to semaglutide: changes in muscle mass, fat distribution, and cellular metabolic health.
The real story is body composition, a complex biological picture that can now be measured precisely through InBody technology. InBody reveals what semaglutide actually does beneath the surface of the scale, not just what you weigh.
What is Semaglutide, and Why Does It Matter for India?
Launched in March 2026, Semaglutide is India’s first DCGI-approved generic semaglutide injection. It is a GLP-1 (glucagon-like peptide-1) receptor agonist, a class of drugs that mimics a naturally occurring gut hormone to regulate blood sugar, suppress appetite, slow gastric emptying, and support weight reduction.
Key facts about Semaglutide:
- Administered once weekly via a pre-filled, disposable subcutaneous injection pen
- Available in 2 mg and 4 mg strengths, each pen delivering a minimum of four weekly doses
- Priced at ₹4,200 per month, making GLP-1 therapy accessible to millions for the first time
- Phase III clinical trial in 312 participants confirmed non-inferior efficacy vs. the innovator drug (Ozempic)
- Day-1 generic entry launched immediately upon patent expiry in India.
101M+ Adults in India living with Type 2 Diabetes (ICMR-INDIAB)
136M Pre-diabetic Indians at high risk of developing the disease
₹4,200 Semaglutide monthly price, the first affordable GLP-1 access in India
India carries one of the world’s heaviest diabetes burdens. Semaglutide changes the affordability equation for GLP-1 therapy, and with that, wider access comes greater clinical responsibility for every physician managing metabolic disease.
When patients begin semaglutide therapy, body weight falls. HbA1c improves. Patients feel encouraged. Clinicians are satisfied. But beneath that number on the scale, a more complex and sometimes concerning biological story is unfolding.
The Problem the Scale Cannot See
Multiple international studies on Semaglutide and related GLP-1 drugs have shown that a significant proportion of total weight lost can come from lean muscle mass, not just body fat. In some trials, up to 25–40% of total weight lost was lean mass, not fat. For older patients or those with existing sarcopenia, this is clinically significant and entirely invisible on a standard weighing scale.
A standard BMI measurement or weighing scale tells you how much someone weighs. An InBody scan tells you what they are made of and whether therapy is working in the right direction.
Key Takeaway: Muscle Loss Drives Metabolic Slowdown
When skeletal muscle declines, resting metabolic rate drops and fat storage becomes more efficient. This explains why some patients on GLP-1 therapy may regain weight after stopping their metabolic engine has downshifted due to lean mass loss, not fat.
What InBody Measures and Why It Matters in GLP-1 Therapy
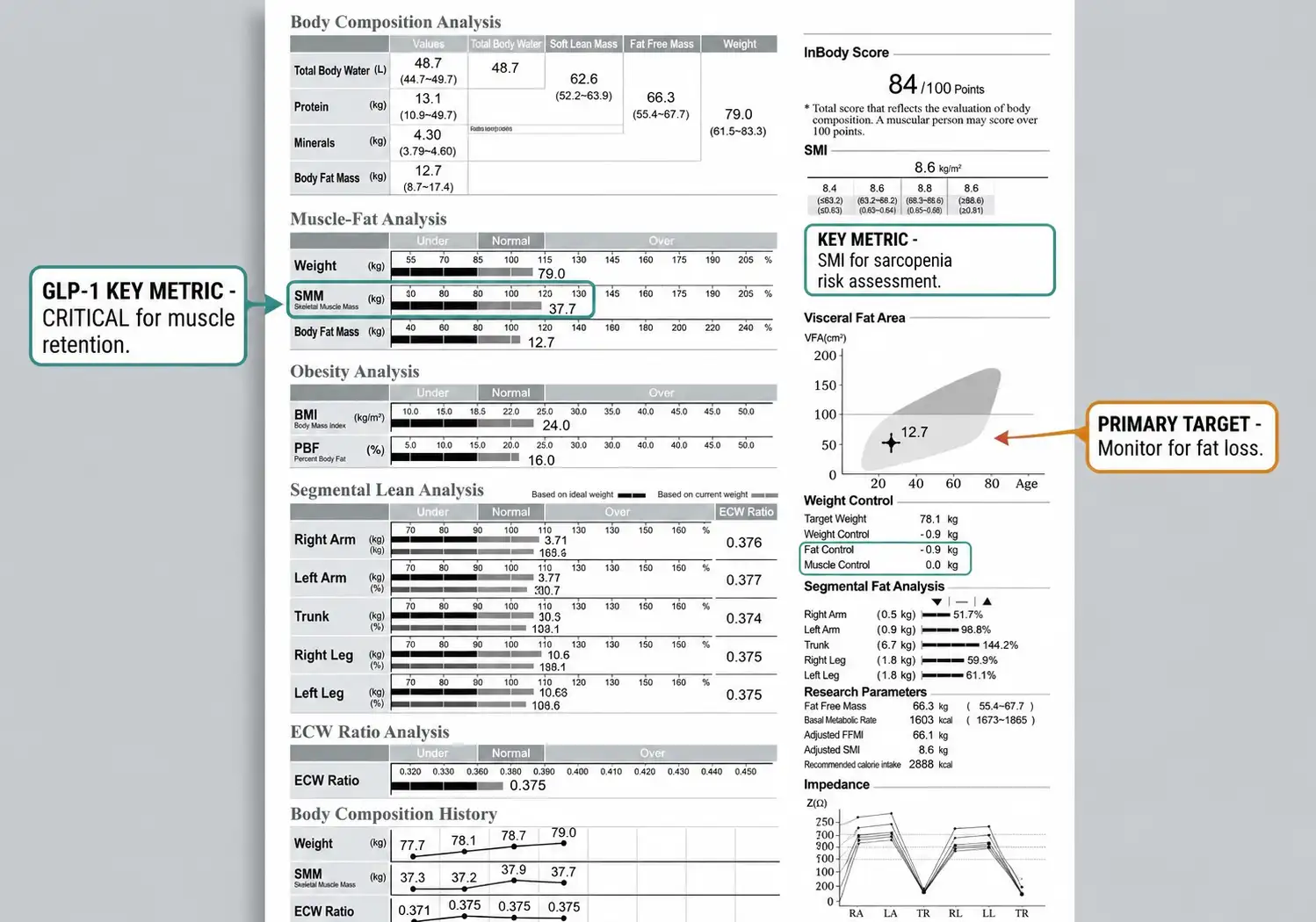
InBody’s bioelectrical impedance analysis (BIA) technology delivers a precise, non-invasive, clinic-ready breakdown of body composition in under 60 seconds. For patients on Semaglutide or any GLP-1 therapy, these are the parameters that matter most:
1. Skeletal Muscle Mass (SMM)
Tracks whether patients are preserving or losing muscle alongside fat. Critical for assessing sarcopenic obesity in older adults on GLP-1 therapy.
2. Body Fat Mass & Percentage
Confirms therapy is driving true fat reduction, not water or lean mass loss. The definitive marker of quality weight loss.
3. Visceral Fat Level
Quantifies metabolically dangerous organ fat directly. Critical for cardiovascular and metabolic risk monitoring on semaglutide therapy.
4. Phase Angle
A marker of cellular health and membrane integrity. Declining phase angle on GLP-1 therapy may signal muscle cell deterioration and trigger intervention.
5. Basal Metabolic Rate (BMR)
Estimated from fat-free mass, not total weight. Tracks whether resting metabolism is being preserved or reduced during therapy.
6. ECW/TBW Ratio
- Extracellular-to-total body water ratio. Elevated values may indicate inflammation or fluid imbalance factors that affect metabolic signalling.
- These measurements explain what is driving weight change during semaglutide therapy, insight that body weight or BMI cannot provide.
Insulin Resistance Visible Through Body Composition
One of the earliest metabolic disruptions in type 2 diabetes is insulin resistance. As muscle cells become less responsive to insulin, glucose redirects toward fat storage rather than energy production. InBody reveals this pattern before it worsens.
Body composition patterns indicating insulin resistance on InBody:
- Increasing body fat mass, especially centrally
- Elevated visceral fat level in the abdomen
- Loss of skeletal muscle mass relative to fat mass
- Declining leg muscle mass, two-thirds of total muscle is in the legs, which is critical for glucose disposal
- Reducing BMR despite stable or rising body weight
InBody’s Segmental Lean Analysis tracks limb muscle changes individually, particularly leg muscle, which plays a crucial role in glucose metabolism and insulin sensitivity.
Why Leg Muscle Matters for Diabetic Patients
Skeletal muscle in the legs is the largest glucose disposal site in the body. When leg muscle mass declines, as can happen on GLP-1 therapy without adequate protein or exercise, insulin sensitivity can paradoxically worsen even as body weight falls. InBody’s segmental analysis is the only way to detect this early.
Visceral Fat: The Hidden Metabolic Risk Semaglutide Targets

Fat tissue is biologically active. As fat accumulates around internal organs, it disrupts metabolic signalling, promotes inflammation, and impairs liver and cardiovascular function. This is precisely what semaglutide is designed to reduce and precisely what InBody quantifies.
Visceral Fat Level is a critical parameter that standard scales and BMI calculations cannot measure. Higher values are associated with:
- Impaired liver metabolism and fatty liver disease
- Poor glucose control and worsening insulin resistance
- Increased cardiovascular and stroke risk
- Chronic low-grade inflammation
Even “normal” weight individuals may show elevated visceral fat on InBody analysis, revealing a hidden metabolic risk that BMI cannot detect. Tracking visceral fat reduction over the course of Semaglutide therapy is one of the most clinically meaningful ways to measure treatment efficacy.
A Suggested Clinical Protocol: Semaglutide + InBody
For clinicians integrating semaglutide therapy into their practice, the following body composition monitoring protocol provides a best-practice framework. It supports better patient outcomes, justifies clinical decisions, and creates an objective data trail for long-term metabolic management.
1. Baseline Scan Before Starting Semaglutide
Conduct an InBody scan before the first dose. Record SMM, body fat %, visceral fat level, phase angle, BMR, and ECW/TBW ratio. This establishes the patient’s true metabolic starting point independent of weight alone.
2. Week 4–6 Check Dose Escalation Phase
Semaglutide begins at a low dose and is escalated over weeks. An InBody scan at 4–6 weeks identifies early disproportionate lean mass loss before it becomes clinically significant, allowing dietary intervention before it progresses.
3. Monthly Monitoring Active Therapy
Monthly scans track the quality of weight loss in real time. If fat mass is declining and SMM is stable or increasing, therapy is on track. If both are declining together, a protein and resistance exercise intervention is warranted immediately.
4. Nutritional Adjustment Protein Optimisation
InBody data combined with dietary assessment allows targeted protein intake recommendations. Adequate protein (1.2–1.6 g/kg body weight) is critical to preserving muscle mass during GLP-1 therapy, especially as appetite suppression reduces overall food intake.
5. 6-Month Outcome Review
Compare the 6-month InBody report to baseline. Calculate net changes in fat mass and lean mass separately. Use this data to counsel patients on maintenance, exercise programming, and whether dose adjustment is warranted.
Scale Weight vs. InBody: What Each One Sees
| Parameter | Weighing Scale | InBody Analysis |
|---|---|---|
| Total body weight | Yes | Yes |
| Body fat mass & percentage | No | Yes |
| Skeletal muscle mass | No | Yes |
| Visceral fat level | No | Yes |
| Basal metabolic rate (BMR) | No | Yes |
| Phase angle (cellular health) | No | Yes |
| ECW/TBW (inflammation marker) | No | Yes |
| Segmental Analysis | No | Yes |
| Distinguishes fat vs. muscle loss | No | Yes |
| Identifies sarcopenic obesity | No | Yes |
| Scan time | Instant | Under 60 seconds |
Deepen Your Expertise in Body Composition Analysis
Healthcare professionals seeking to master metabolic assessment and body composition science can gain hands-on training at InBody Academia, an intensive learning experience covering clinical interpretation, metabolic risk identification, and practical application of InBody data in real patient care.
Tracking Metabolic Adaptation Over a Course of Semaglutide
As semaglutide therapy progresses, the body may reduce energy expenditure beyond what size alone would predict, a phenomenon called adaptive thermogenesis. This is especially pronounced when lean mass is lost alongside fat.
Serial InBody assessments reveal adaptation through:
- Falling BMR despite maintained or reduced weight
- Muscle loss with simultaneous fat regain after stopping therapy
- Slower weight loss plateau despite continued medication
- Declining phase angle indicates cellular health deterioration
Tracking these trends is essential for understanding why weight regain is common after stopping GLP-1 therapy and why concurrent muscle preservation strategies are non-negotiable in any complete semaglutide management programme.
Key Takeaway: Early Detection Through Body Composition
Body composition changes appear before weight gain. Tracking BMR, muscle mass, visceral fat, phase angle, and hydration markers reveals metabolic problems months or years before traditional clinical measures would detect them.
The Complete Metabolic Picture: Beyond Body Weight
Body weight is only the surface outcome of deeper metabolic processes: energy partitioning, muscle-to-fat balance, visceral fat burden, cellular health, and hydration status. InBody translates these processes into measurable parameters, allowing clinicians to move from assumptions into objective insight.
Body composition analysis with InBody provides:
- Accurate BMR measurement for personalised nutrition planning during GLP-1 therapy
- Visceral fat assessment for metabolic disease risk evaluation at each visit
- Muscle mass tracking to prevent metabolic slowdown and sarcopenia
- Phase angle monitoring for cellular health across the treatment course
- Water distribution analysis for detecting inflammation and fluid imbalance
This comprehensive metabolic assessment is alone, and it is now the clinical standard of care in progressive metabolic medicine globally.
Frequently Asked Questions
Q. What is Semaglutide and how does it work?
Semaglutide is India’s first DCGI-approved generic injection. It is a GLP-1 receptor agonist that mimics a natural gut hormone, improving blood sugar control, reducing appetite, and supporting weight loss in adults with type 2 diabetes. It is administered once weekly via a pre-filled subcutaneous pen at ₹4,200 per month.
Q. Does semaglutide cause muscle loss?
Yes, without proper nutrition and resistance training, part of the weight loss can come from muscle mass.
Q. How can I lose fat without losing muscle?
By maintaining adequate protein intake, strength training, and monitoring body composition.
Q. Is weight loss the same as fat loss?
No, weight loss includes both fat and muscle loss, which is why body composition tracking is important.
Q. How does an InBody scan help during weight loss?
It differentiates between fat mass and muscle mass changes, helping ensure healthy fat loss.
Conclusion: India’s GLP-1 Era Needs a Body Composition Partner
Semaglutide‘s launch marks a turning point in how India manages metabolic disease. For the first time, a clinically validated, DCGI-approved, affordable GLP-1 therapy is within reach for millions of Indians who need it. That is genuinely significant progress.
But the clinical responsibility that comes with wider GLP-1 adoption is equally significant. Weight loss is not the same as fat loss. A falling number on the scale is not always a sign of better metabolic health, especially if what is falling includes the muscle that patients need for long-term metabolic function, glucose disposal, and quality of life.
Body composition analysis with InBody translates the promise of semaglutide therapy into measurable, manageable, objective outcomes at every step of the treatment journey.
As India enters its GLP-1 era, the clinics that lead won’t just prescribe the injection. They’ll measure what truly changes inside.
Medical Disclaimer: This blog is intended for healthcare professionals and is for informational and educational purposes only. It does not constitute medical advice, diagnosis, or treatment recommendations. Semaglutide is a prescription medication; all clinical decisions must be made by qualified healthcare providers based on individual patient assessment. InBody results should be interpreted in conjunction with a full clinical evaluation.
References & Further Reading
- DCGI / CDSCO Approval Tracking: Central Drugs Standard Control Organisation (CDSCO) — Used to verify the generic drug approval registry for 2026.
- ICMR-INDIAB Study (PIB): Prevalence of Diabetes and Prediabetes in India (101 Million Adults)
- The Lancet Diabetes & Endocrinology: Metabolic non-communicable disease health report of India: the ICMR-INDIAB national cross-sectional study
- The STEP 1 Trial (NEJM): Once-Weekly Semaglutide in Adults with Overweight or Obesity — The primary clinical trial demonstrating weight loss efficacy.
- Lean Mass Loss Review (Obesity Reviews): A systematic review of the effect of semaglutide on Lean Mass (Bikou O, et al.)
- Muscle Mass & Metabolic Rate (AJCN): Skeletal muscle mass and distribution in 468 men and women (Janssen I, et al., 2000)
- InBody USA White Paper: Understanding Body Fat & Muscle Categories — Explains why BMI is insufficient and how Direct Segmental Multi-frequency BIA (DSM-BIA) works.
- Healthline – Muscle Loss & Ozempic/Wegovy: What to Know About Muscle Loss While Taking Semaglutide



