Your kidneys can lose 60% of their function before you feel a single symptom. No pain, swelling and no change in urine. Just silent, progressive damage accumulating year after year until a blood test, a cardiac event, or a hospitalisation finally reveals a disease that has been advancing for a decade. This is why chronic kidney disease (CKD) is one of the most dangerous conditions in medicine and one of the least discussed in India.
The numbers are alarming. The SEEK-India multi-centre study found a CKD prevalence of 17.2% across its cohort, meaning approximately 1 in 6 Indians has some form of kidney disease. India’s ESRD incidence rate is estimated at 229 per million population, with over 100,000 new patients entering renal replacement programmes every year. Between 2001 and 2013, the proportion of deaths attributable to kidney failure in India rose by 38%. In this article, we examine the 7 most dangerous clinical reasons kidney disease kills silently and the evidence-based actions that actually stop it.
| 17.2% CKD prevalence in the SEEK-India multi-centre cohort approximately 1 in 6 Indians (BMC Nephrology) | 38% Rise in deaths attributable to kidney failure in India between 2001–03 and 2010–13 (ICKD, CKJ 2022) | 90% CKD patients unaware of their diagnosis until Stage 3 or beyond when irreversible damage is established | 229 pmp India’s ESRD incidence rate (per million population) over 100,000 new patients annually |
Why Kidney Disease Earns the ‘Silent Killer’ Label
The kidneys have extraordinary compensatory capacity. A healthy adult has approximately 2 million nephrons, the functional filtering units of the kidney. When nephrons are damaged and lost, the remaining ones hypertrophy and work harder. This compensation is so effective that patients can lose 60–70% of total nephron mass before their eGFR drops below 60 mL/min/1.73m², the threshold for Stage 3 CKD. And Stage 3 CKD, in most patients, produces no symptoms whatsoever.
The first noticeable sign of kidney disease in many Indian patients is not fatigue, not oedema, and not a change in urine output. It is a cardiovascular event, a heart attack, stroke, or hospitalisation for heart failure that is the downstream consequence of years of undetected CKD silently driving hypertension, dyslipidaemia, anaemia, and fluid overload. By the time the diagnosis is made, the window for meaningful disease modification has often passed.
THE INDIAN CKD PARADOX
India’s CKD burden is compounded by a structural gap: patients seek medical attention when they are symptomatic, but CKD produces symptoms only in its later stages. The 2025 ICMR-led meta-analysis of community-based studies confirmed wide regional variation in CKD prevalence, with rural and semi-urban populations carrying a disproportionate burden. CKD in India is a disease of late presentation, underdiagnosis, and missed prevention windows. All three are addressable with the right clinical tools.
Reason 1: No Symptoms Until Serious Damage Is Already Done
The absence of symptoms is the defining clinical feature of CKD in its early stages. Fatigue, nausea, loss of appetite, oedema, and reduced urine output,, the classic symptoms most patients associate with kidney disease typically appear only in Stage 4 or Stage 5 CKD, when eGFR has fallen below 29 mL/min/1.73m². At this stage, renal replacement therapy is weeks to months away, not years.
The ICKD (Indian Chronic Kidney Disease) Study confirmed this clinical reality. Two-thirds of participants in the ICKD cohort were male, reflecting not a true gender difference in CKD risk, but a systematic healthcare presentation barrier; women with early CKD in India are less likely to present for evaluation until symptoms are severe. This means that for a substantial proportion of India’s female population with CKD, the disease is progressing in complete clinical silence, undetected by any current screening programme.
CLINICAL IMPLICATION
Do not wait for symptoms to screen for CKD. The KDIGO 2024 guidelines recommend annual eGFR and urine albumin-to-creatinine ratio (uACR) testing for all patients with diabetes, hypertension, or a family history of kidney disease, regardless of symptoms. Early CKD is a laboratory diagnosis, not a clinical one. Any patient with a uACR above 30 mg/g on two separate measurements has CKD by KDIGO definition, irrespective of eGFR.
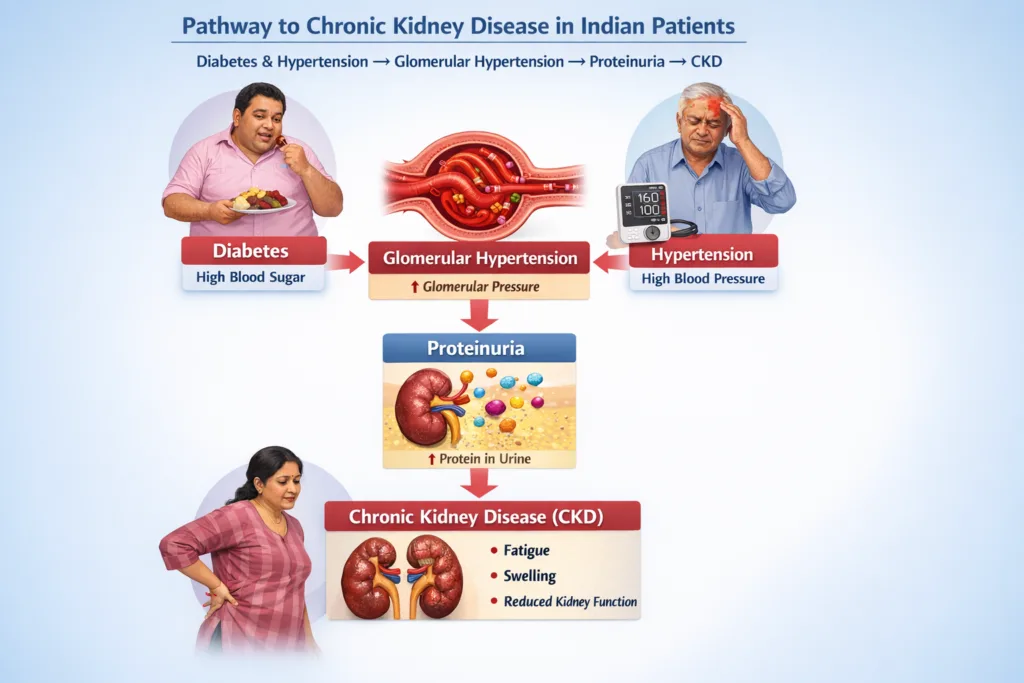
Reason 2: Diabetes and Hypertension Are Silently Destroying the Kidneys of Most Indians
The SEEK-India study found that hypertension was present in 43.1% of its CKD cohort and diabetes in 18.8%. These are not coincidences; they are the two primary drivers of CKD in India, causing diabetic nephropathy and hypertensive nephrosclerosis respectively. Both destroy the kidneys through the same final common pathway: glomerular hypertension leading to progressive proteinuria, fibrosis, and nephron loss.
India has 101 million people with type 2 diabetes and approximately 220 million with hypertension. A proportion of these patients already have CKD,, and most do not know it. The CURES study (Chennai Urban Rural Epidemiology Study) found that in urban South Indians with diabetes, 26.9% already had diabetic nephropathy. These patients are not presenting to nephrologists. They are presenting to general practitioners and diabetologists for glucose management and their kidney function is not being serially monitored.
KEY INSIGHT
For every patient you see with T2DM or hypertension, kidney damage may already be progressing. The standard of care is annual eGFR + uACR in all T2DM patients from diagnosis, and in all hypertensive patients within 12 months of diagnosis. Starting RAAS blockade (ACE inhibitor or ARB) at the first sign of microalbuminuria reduces CKD progression by 30–45% in diabetic nephropathy, but only if the window is identified in time.
Reason 3: Cardiovascular Disease and CKD Are Locked in a Deadly Feedback Loop
CKD is not merely a kidney problem. It is one of the most powerful independent risk factors for cardiovascular disease, producing a hazard ratio for cardiovascular events between 1.4 and 3.4, depending on CKD stage and the presence of proteinuria. The mechanism is multi-pathway: CKD drives uraemic cardiomyopathy, endothelial dysfunction, vascular calcification, accelerated atherosclerosis, anaemia-related cardiac strain, and fluid overload-driven left ventricular hypertrophy simultaneously.
The ICKD study confirmed that CKD is a major cardiovascular disease risk factor and a leading cause of premature deaths and disability-adjusted life years in India. Most CKD patients in India do not die from kidney failure; they die from cardiovascular events long before reaching ESRD. This means that treating CKD as purely a renal disease and failing to aggressively manage its cardiovascular consequences systematically underestimates the clinical urgency of early intervention.
THE BODY COMPOSITION LINK
Fluid overload driven by impaired ECW regulation as eGFR declines is a primary driver of cardiovascular mortality in CKD. A rising ECW/TBW ratio, measurable with InBody BIA at every clinic visit, is both an early marker of deteriorating fluid regulation and an independent predictor of cardiovascular events in CKD patients detectable months before clinical symptoms of volume overload appear.
Reason 4: Proteinuria: The Warning Sign That Is Almost Always Missed
Proteinuria, the presence of excess protein in the urine, is the earliest detectable marker of kidney damage in most CKD aetiologies. It precedes eGFR decline by years in diabetic nephropathy and hypertensive CKD. A urine albumin-to-creatinine ratio (uACR) above 30 mg/g on two separate measurements over at least three months constitutes albuminuria and defines CKD even in patients with completely normal eGFR.
The Delhi-Chennai cohort study found that 80% of participants with CKD had albuminuria alone, meaning their eGFR was still in the normal range. These patients have Stage 1 or Stage 2 CKD by KDIGO criteria, and they represent the most important early intervention opportunity in nephrology. Yet in standard primary care practice across India, uACR is not routinely measured. Dipstick urinalysis, which misses microalbuminuria by design, remains the most commonly performed urine test in outpatient settings.
CLINICAL IMPLICATION
Replace dipstick urinalysis with spot urine albumin-to-creatinine ratio (uACR) for all high-risk patients. A uACR between 30–300 mg/g defines microalbuminuria (moderately increased albuminuria, Stage A2). A uACR above 300 mg/g defines macroalbuminuria (severely increased, Stage A3). Every patient with T2DM, hypertension, or obesity should have a uACR at least annually. This single test change will identify CKD years before eGFR decline begins.
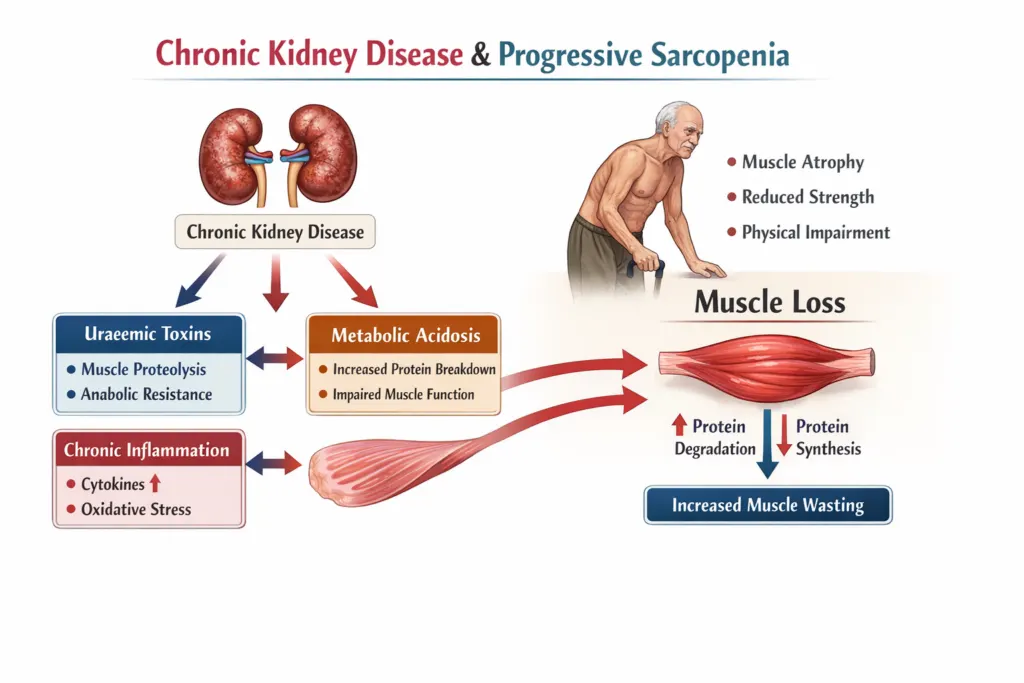
Reason 5: Sarcopenia Is Silently Accelerating in Every CKD Patient
Chronic kidney disease is one of the most potent drivers of skeletal muscle loss in medicine. Uraemic toxins directly suppress muscle protein synthesis. Metabolic acidosis activates the ubiquitin-proteasome pathway, accelerating muscle catabolism. Chronic inflammation through elevated IL-6 and TNF-α drives inflammatory myopathy. Reduced physical activity consequent to fatigue and restricted dietary protein further accelerates the process. The result is progressive sarcopenia that begins in early CKD and worsens with declining eGFR.
The clinical consequences are profound. Sarcopenia in CKD patients is independently associated with hospitalisation, falls, fractures, and mortality. It also creates a dangerous diagnostic trap: as skeletal muscle mass declines, serum creatinine used in all eGFR equations falls. A patient whose creatinine drops from 1.2 to 0.9 mg/dL may appear to be improving. But if this drop reflects muscle loss rather than improved filtration, eGFR is being systematically overestimated, and the true rate of CKD progression is being missed.
THE CREATININE TRAP IN CKD
Serum creatinine is a product of muscle metabolism. In sarcopenic CKD patients, falling creatinine may reflect muscle loss rather than improved kidney function. InBody BIA directly measures skeletal muscle mass in kilograms, independent of creatinine or eGFR. Serial InBody assessment in CKD patients simultaneously tracks muscle preservation and fluid status, providing the clinical data that serum creatinine alone cannot deliver. The Indian Consensus on Sarcopenia (2025) recommends BIA for all patients with CKD 3 and above.
Reason 6: Fluid Overload Is Building Long Before Dialysis Begins
Fluid overload is not exclusively a dialysis problem. As eGFR declines from Stage 3 onwards, the kidneys’ ability to regulate sodium and water balance progressively deteriorates. The extracellular fluid compartment expands. The ECW/TBW ratio rises. Blood pressure climbs. The left ventricle enlarges. All of this occurs in pre-dialysis CKD patients who may have no visible oedema, no measurable weight gain from day to day, and no clinical flags to alert their physician.
The cardiovascular mortality that kills most CKD patients before they reach dialysis is substantially driven by this progressive, clinically invisible fluid overload. A rising ECW/TBW ratio in a CKD Stage 3 patient detectable with InBody BIA at a routine nephrology appointment represents the early stage of the same process that produces the 69.6% 30-month mortality seen in dialysis patients with post-dialysis ECW/TBW above 0.5023. The window to intervene is now not when dialysis begins.
WHAT InBody S10 DETECTS IN PRE-DIALYSIS CKD
ECW/TBW ratio trend: rising values indicate progressive volume overload before clinical symptoms
Overhydration index (OH in litres): quantifies the absolute fluid burden above euvolaemia
Skeletal muscle mass trend: identifies sarcopenic progression concurrent with fluid assessment
Fat tissue index: tracks body composition change as dietary protein restriction is introduced
These four parameters together provide a complete metabolic picture of CKD progression that serum creatinine, eGFR, and body weight alone cannot deliver.
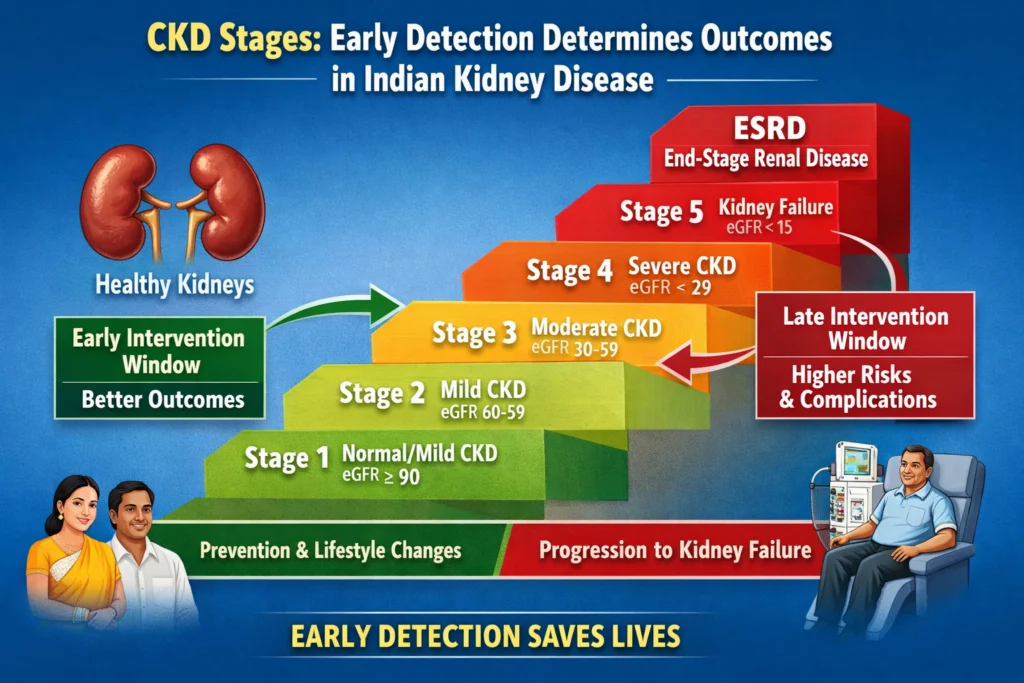
Reason 7: Late Presentation Means Most Indian CKD Patients Miss Their Best Treatment Window
The Million Death Study a nationally representative mortality survey, found that the highest burden of age-standardised renal deaths in India was concentrated in the 45–69 age group. This represents working-age adults at the peak of their economic productivity. Yet data from the ICKD study and SEEK-India consistently confirm that Indian patients seek medical attention for kidney disease when they are symptomatic, by which time the disease is advanced, treatment options are limited, and outcomes are significantly worse than for early-detected CKD.
The 2025 ICMR-led meta-analysis confirmed that CKD in India often goes undetected until its advanced stages. This is not a patient education problem alone; it is a healthcare system problem. Primary care physicians who manage the bulk of India’s diabetic and hypertensive patients are the frontline of CKD detection. The tools for early detection are simple, inexpensive, and available. The practice gap is the barrier.
WHAT ‘STOPPING IT’ ACTUALLY REQUIRES
The interventions that have the strongest evidence for slowing CKD progression are: tight BP control (target <130/80 mmHg), RAAS blockade at first microalbuminuria, SGLT-2 inhibitors (proven renoprotective independent of glycaemic effect CREDENCE, DAPA-CKD, EMPA-KIDNEY trials), dietary protein restriction in CKD Stage 4–5, smoking cessation, and early anaemia management. To implement these interventions at the right time, you need to detect CKD early. And to detect it early, you need to measure more than creatinine.
How to Actually Stop It: The Evidence-Based Action Plan
Stopping kidney disease requires action at three levels simultaneously: detecting it early, treating the upstream causes aggressively, and monitoring both kidney function and body composition to confirm that intervention is working. Here is the evidence-based framework:
| Action | What to Do | Evidence Base |
|---|---|---|
| Screen High-Risk Patients Annually | eGFR + uACR in all T2DM, hypertension, and obesity patients from diagnosis, not from symptoms | KDIGO 2024 CKD Guidelines; SEEK-India; CURES Study |
| Start RAAS Blockade Early | ACE inhibitor or ARB at first microalbuminuria (uACR above 30 mg/g). Target BP below 130/80 mmHg | RENAAL, IDNT, ADVANCE trials 30–45% CKD progression reduction |
| Add SGLT-2 Inhibitor | For CKD patients with or without T2DM proven renoprotective independent of glucose lowering | CREDENCE, DAPA-CKD (dapagliflozin), EMPA-KIDNEY (empagliflozin) 2022–2023 |
| Monitor Body Composition | Serial InBody BIA at every nephrology visit, ECW/TBW ratio, SMM, overhydration index, FTI. | Indian Consensus on Sarcopenia 2025; KDOQI Nutrition Guidelines CKD 5D |
| Manage Protein Intake | 0.6–0.8 g/kg/day in CKD 4–5 (pre-dialysis). 1.2–1.5 g/kg/day on dialysis. Track with InBody SMM | ICMR-NIN 2024; Indian Consensus on Sarcopenia 2025 |
| Correct Anaemia Early | Target Hb 10–12 g/dL in CKD anaemia. Reduces cardiac strain and improves quality of life | KDIGO Anaemia in CKD Guidelines 2024 |
Real Case: The Patient Whose Kidney Disease Was ‘Hidden’ in a Normal Creatinine
A 54-year-old woman with a 12-year history of T2DM and hypertension attended a corporate health check in Mumbai. Her serum creatinine was 0.85 mg/dL, well within the normal reference range. Her treating physician had reviewed her blood results 3 months earlier and noted no concerns. She had no symptoms.
Her InBody scan at the health check revealed Skeletal Muscle Mass 2.4 kg below the population norm for her age and height. Her referring physician, alerted by the below-normal muscle mass, subsequently checked a urine albumin-to-creatinine ratio, which had never been tested in her 12 years of diabetes management. The result was 68 mg/g Stage A2 albuminuria. A formal eGFR using CKD-EPI was calculated at 54 mL/min/1.73m² for Stage 3a CKD. Her creatinine appeared normal because her low muscle mass was producing less creatinine than expected for her actual kidney function.
She began RAAS blockade, SGLT-2 inhibitor therapy, and protein-adjusted dietary management. At her 6-month review, her uACR had reduced from 68 to 31 mg/g, a 54% reduction, confirming treatment response. Her eGFR had stabilised. The InBody scan showed:
| ↓54% uACR reduction (68 → 31 mg/g) at 6 months | Stable eGFR, no further decline during treatment period | ↓0.018 ECW/TBW ratio improvement at 6 months | +1.1 kg Skeletal Muscle Mass gained with protein management |
Her creatinine had remained within the normal range throughout. Without the InBody-prompted uACR test, her CKD would have gone undetected for years, progressing silently toward dialysis-dependent ESRD while appearing normal on every standard blood panel.

Frequently Asked Questions
Q. How common is kidney disease in India?
The SEEK-India multi-centre study found a CKD prevalence of 17.2% approximately 1 in 6 Indians. India’s ESRD incidence rate is 229 per million population, with over 100,000 new patients entering renal replacement programmes annually. The 2025 ICMR-led meta-analysis confirmed wide regional variation, with higher prevalence in rural populations and regions with agricultural nephrotoxin exposure. CKD deaths in India rose by 38% between 2001–03 and 2010–13, and this trajectory has not reversed.
Q. What are the earliest detectable signs of kidney disease?
The earliest detectable signs before symptoms develop are microalbuminuria (uACR above 30 mg/g), a declining eGFR trend across serial measurements, and rising blood pressure. Body composition changes also precede symptoms: declining skeletal muscle mass, rising ECW/TBW ratio, and falling intracellular water, all measurable with InBody BIA, indicate metabolic deterioration in CKD patients before clinical symptoms emerge. The creatinine trap is particularly important: falling creatinine in a sarcopenic CKD patient may reflect muscle loss rather than improving kidney function, causing systematic overestimation of true eGFR.
Q. What is the connection between kidney disease and body composition?
CKD causes progressive sarcopenia through uraemic toxin-driven muscle catabolism, metabolic acidosis, and chronic inflammation. It simultaneously drives fluid overload through impaired ECW regulation as eGFR declines. Both processes worsen outcomes independently: sarcopenia is associated with hospitalisation and mortality in CKD patients, while fluid overload drives the cardiovascular mortality that kills most CKD patients before they reach dialysis. InBody BIA simultaneously measures skeletal muscle mass, ECW/TBW ratio, and overhydration index, enabling clinicians to track both disease trajectories at every visit, from CKD Stage 3 through to dialysis.
Key Takeaways
- 17.2% of Indians have CKD, and most are unaware until Stage 3 or beyond, when irreversible damage has already been established, and treatment options are substantially reduced.
- The 7 dangerous reasons CKD kills silently, no symptoms, diabetes and hypertension as silent drivers, the cardiovascular feedback loop, missed proteinuria, silent sarcopenia, pre-dialysis fluid overload, and late presentation all have one common solution: earlier, more comprehensive screening that goes beyond serum creatinine alone.
- 80% of patients with CKD in the Delhi-Chennai cohort had albuminuria alone with normal eGFR. These Stage 1–2 CKD patients are the most important early intervention opportunity in nephrology, and they are currently being missed by dipstick urinalysis.
- Falling creatinine does not always mean improving kidney function in sarcopenic CKD patients; it may reflect muscle loss. InBody BIA directly measures skeletal muscle mass, detecting this diagnostic trap and providing the complete metabolic picture that creatinine alone cannot deliver.
- SGLT-2 inhibitors (dapagliflozin, empagliflozin), RAAS blockade, and tight BP control, started when proteinuria is first detected, reduce CKD progression by 30–45%. These interventions only work if CKD is detected early enough to apply them.
- InBody BIA simultaneously measures ECW/TBW ratio, overhydration index, and skeletal muscle mass at every nephrology visit, tracking both the fluid overload and sarcopenia trajectories that serum markers cannot capture, from CKD Stage 3 through to dialysis.
Detect What Creatinine Cannot Track What eGFR cannot.
InBody body composition assessment provides the fluid, muscle, and nutritional data that CKD management requires beyond standard blood panels in under 5 minutes at the bedside, at every visit. Used in nephrology units, dialysis centres, and metabolic clinics across India.

Find an InBody Scanner Near You
References & Further Reading
- Rajapurkar MM et al. SEEK-India: Epidemiology and risk factors of CKD in India. BMC Nephrology. 2013;14:114.
- Kumar V, Yadav AK et al. Indian Chronic Kidney Disease (ICKD) study: baseline characteristics. Clinical Kidney Journal. 2022;15(1):60–69.
- Talukdar R et al. CKD Prevalence in India: Systematic Review and Meta-Analysis 2011–2023. Nephrology. 2025;30:e14420.
- Siddiqui NY et al. Prevalence of CKD in Delhi and Chennai with CVD projections. PMC4490055.
- Jha V. CKD in India: from a resident physician’s perspective. PMC2660492.
- Kalra S et al. Indian Consensus on Sarcopenia including CKD 3–5D recommendations. Int J Gen Med. 2025;18:1731–1745.
- KDIGO 2024 CKD Clinical Practice Guidelines. Kidney International. 2024.
- Perkovic V et al. CREDENCE trial — canagliflozin and renal outcomes in T2DM and nephropathy. NEJM. 2019;380:2295–2306.
